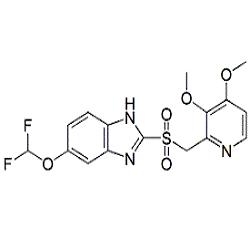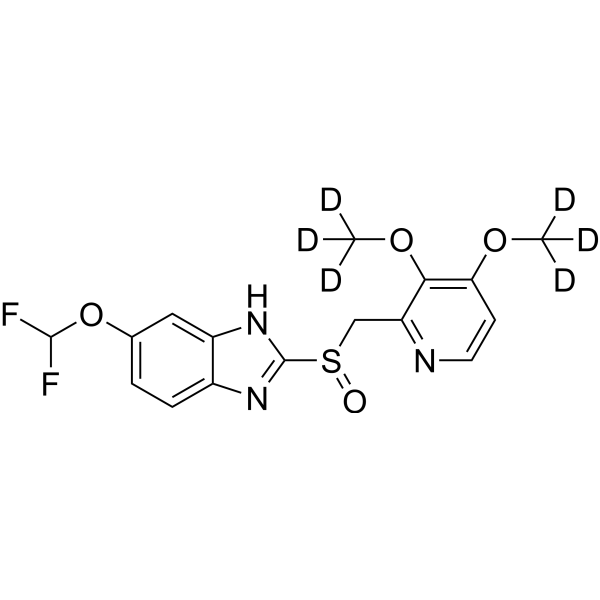
Pantoprazole Related Compound B pharmaceutical secondary standard, certified reference material | 102625-64-9

Environmentally Benign and Facile Process for the Synthesis of Pantoprazole Sodium Sesquihydrate: Phase Transformation of Pantoprazole Sodium Heterosolvate to Pantoprazole Sodium Sesquihydrate | ACS Omega

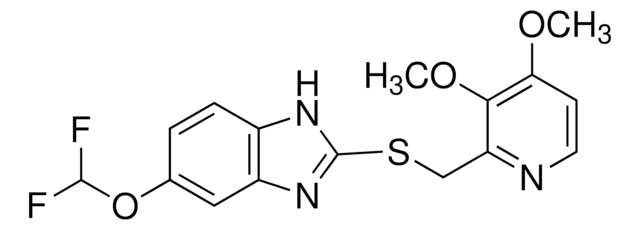
Pantoprazole EP Impurity B|CAS.No-102625-64-9|Pantoprazole EP Impurity B, Pantoprazole USP Related Compound B|Impurity Standards, Metabolites

Formulation and Evaluation of Pantoprazole Sodium EC Tablets: Pantoprazole Sodium Delayed Release Tablets: Unnisa Begum, Rahmath: 9783330038776: Amazon.com: Books
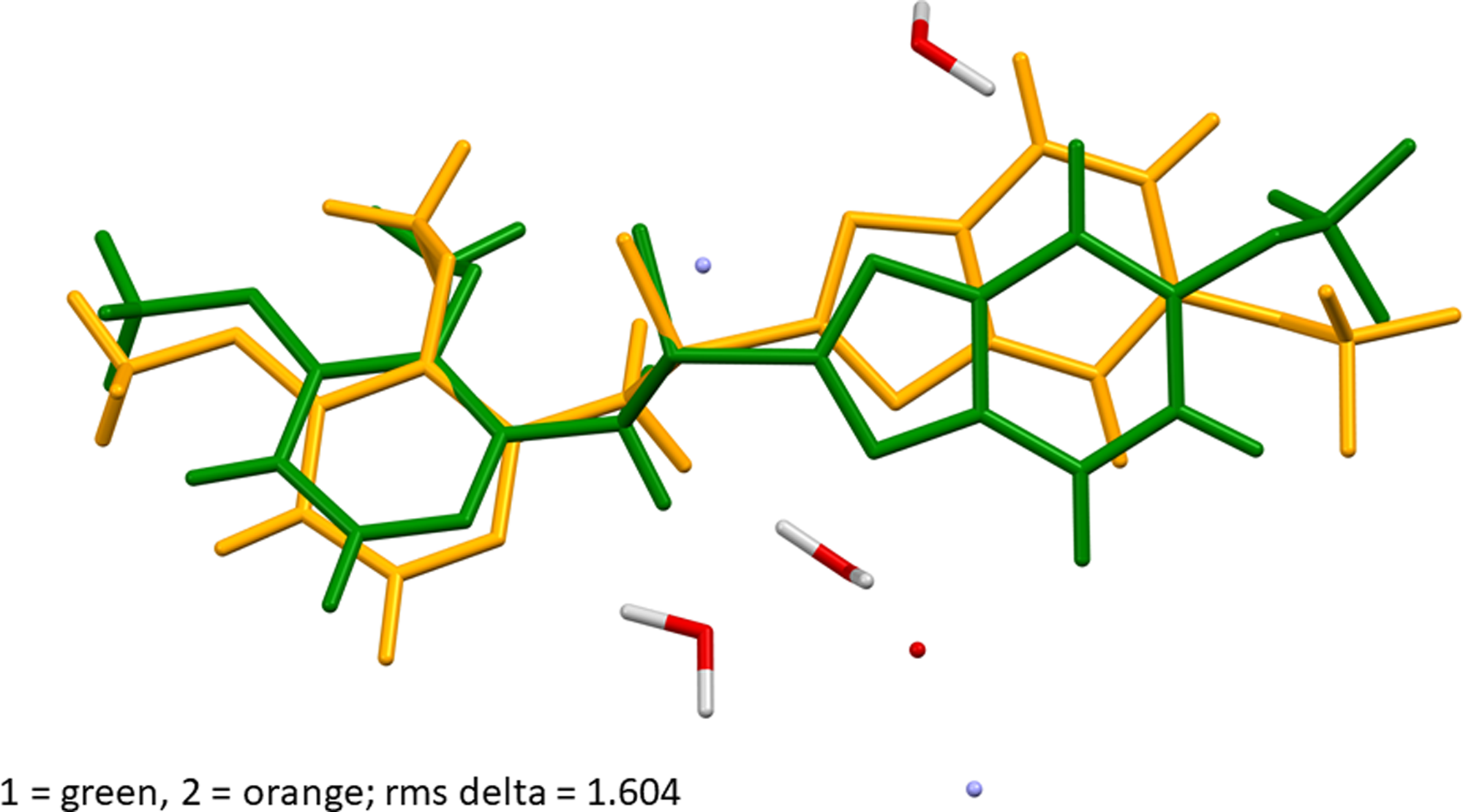
Crystal structure of pantoprazole sodium sesquihydrate Form I, C16H14F2N3O4SNa(H2O)1.5 | Powder Diffraction | Cambridge Core
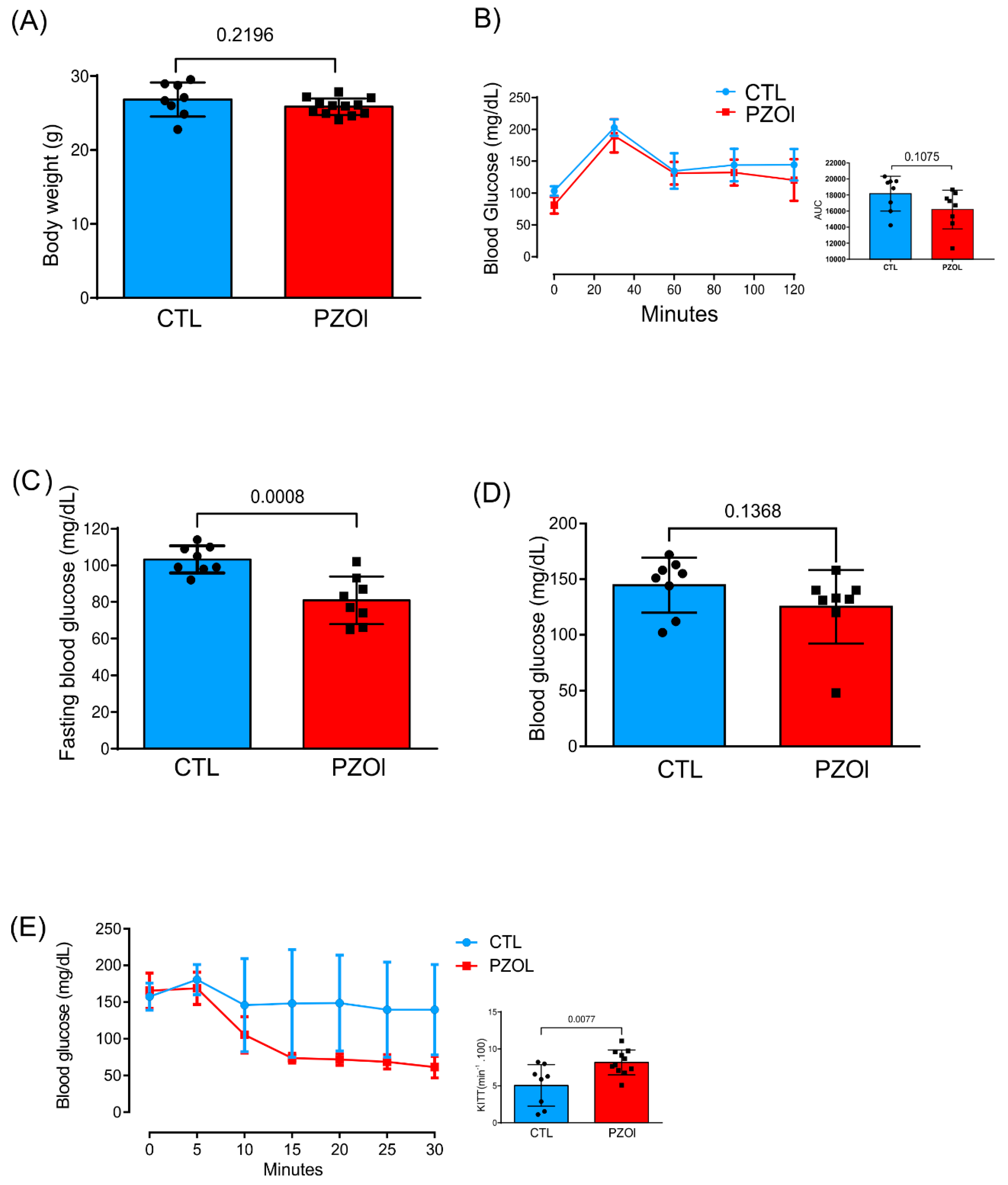
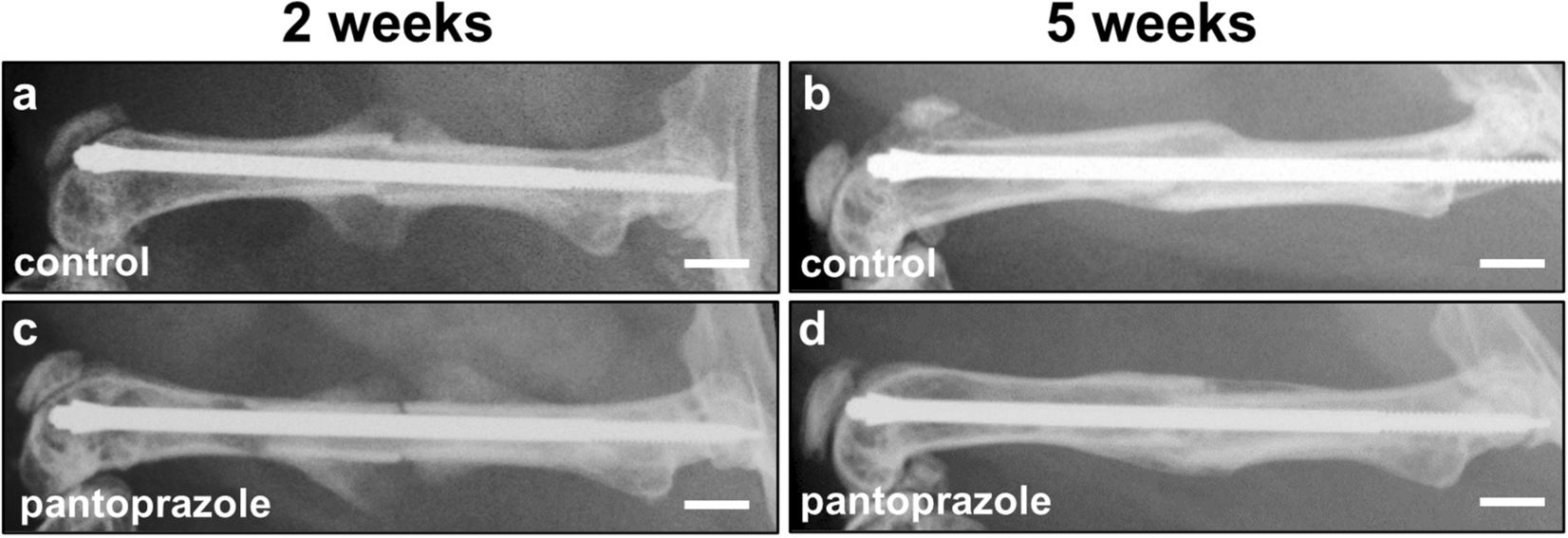
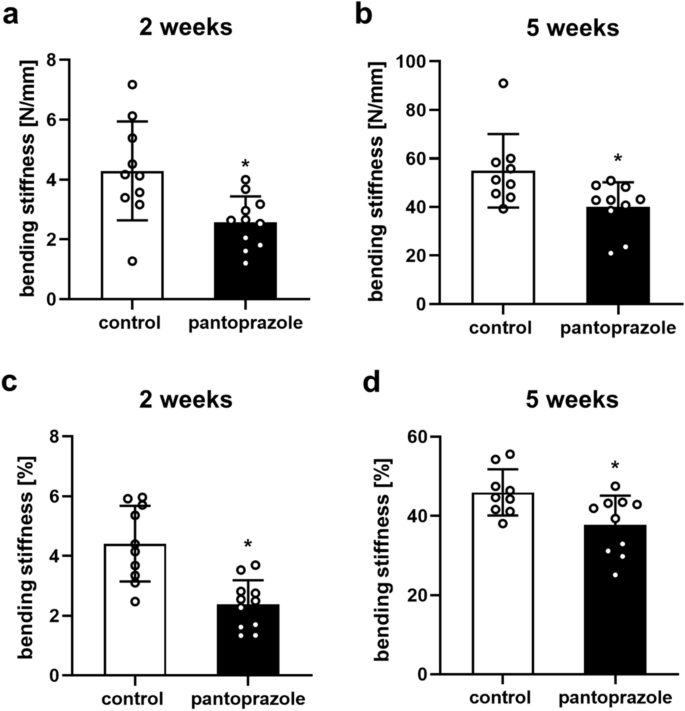
IJMS | Free Full-Text | Proton Pump Inhibitor Pantoprazole Modulates Intestinal Microbiota and Induces TLR4 Signaling and Fibrosis in Mouse Liver

Environmentally Benign and Facile Process for the Synthesis of Pantoprazole Sodium Sesquihydrate: Phase Transformation of Pantoprazole Sodium Heterosolvate to Pantoprazole Sodium Sesquihydrate | ACS Omega
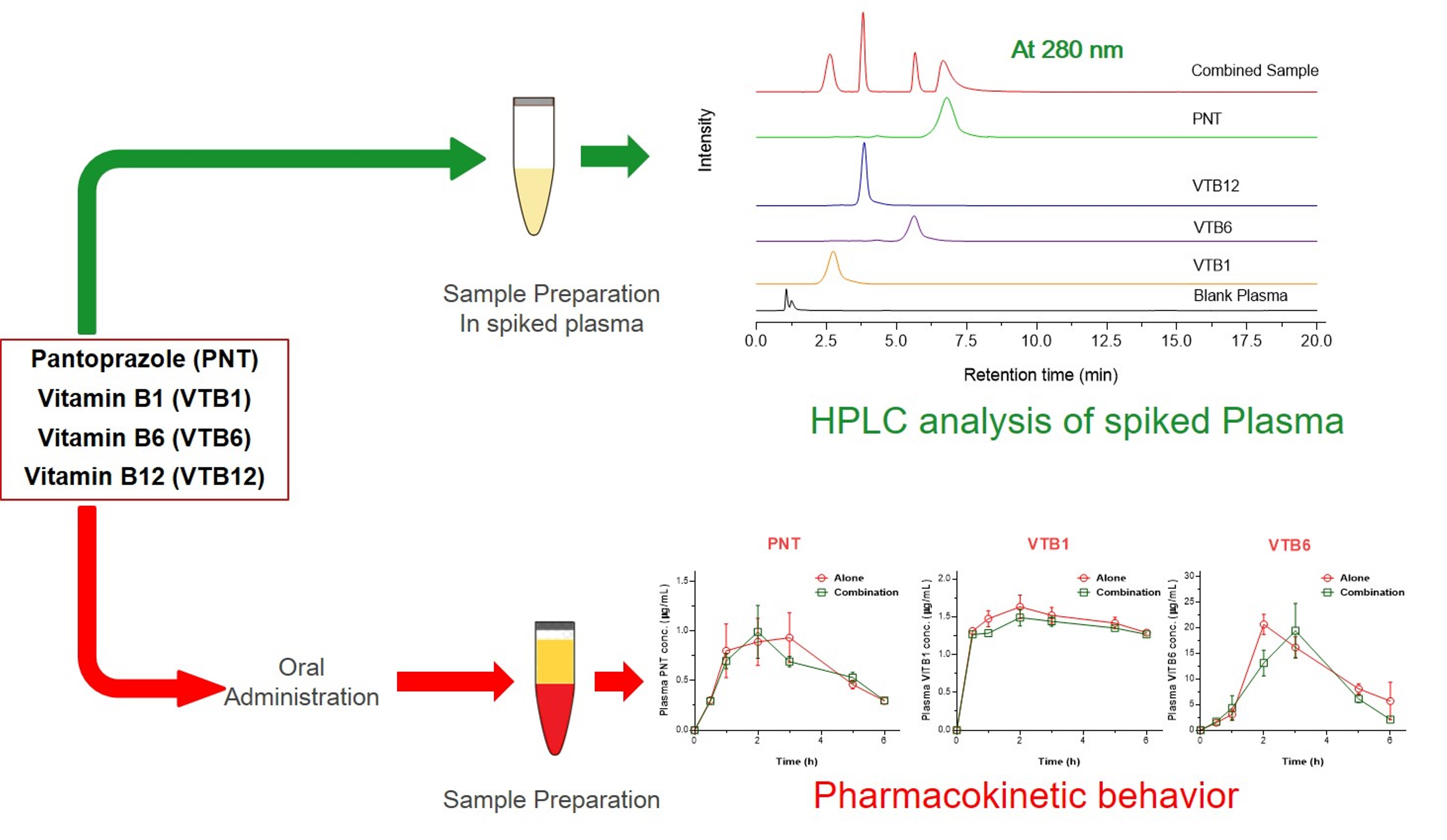
Separations | Free Full-Text | Simultaneous Pharmacokinetic Evaluation of Pantoprazole and Vitamin B Complex for Assessing Drug–Drug Interactions in Healthy Bangladeshi Adults by a Newly Developed and Validated HPLC Method
![102625-64-9 | Pantoprazole Sulfide | 5-(Difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridinyl)methyl]thio]-1H-benzimidazole; Pantoprazole Thioether; Pantoprazole EP Impurity B; | C₁₆H₁₅F₂N₃O₃S | TRC 102625-64-9 | Pantoprazole Sulfide | 5-(Difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridinyl)methyl]thio]-1H-benzimidazole; Pantoprazole Thioether; Pantoprazole EP Impurity B; | C₁₆H₁₅F₂N₃O₃S | TRC](https://www.trc-canada.com/prod-img/P183020.png)