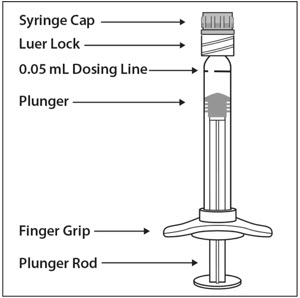
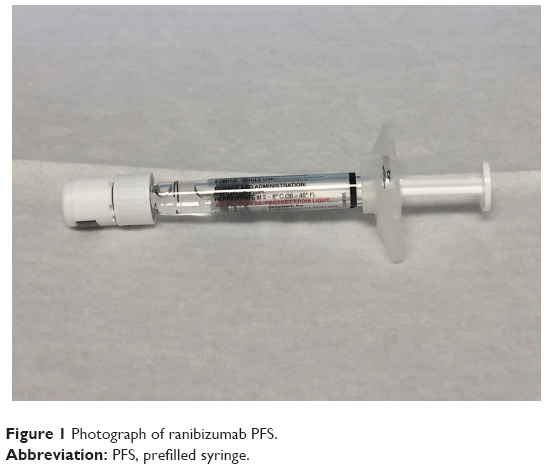
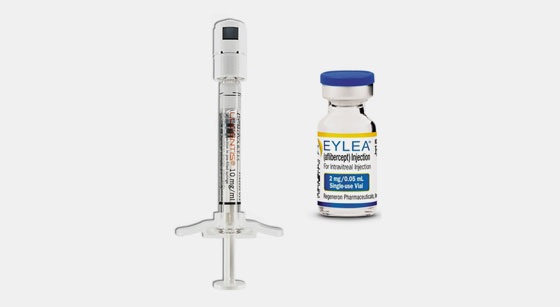
UNITED STATES INTERNATIONAL TRADE COMMISSION WASHINGTON, D.C. In the Matter of CERTAIN PRE-FILLED SYRINGES FOR INTRAVITREAL INJE
WVU Eye Institute retinopathy of prematurity clinical trial results in FDA approval | School of Medicine | West Virginia University

Drug delivery to the eye: overcoming the challenges of intravitreal prefilled syringes | Cambridge Consultants
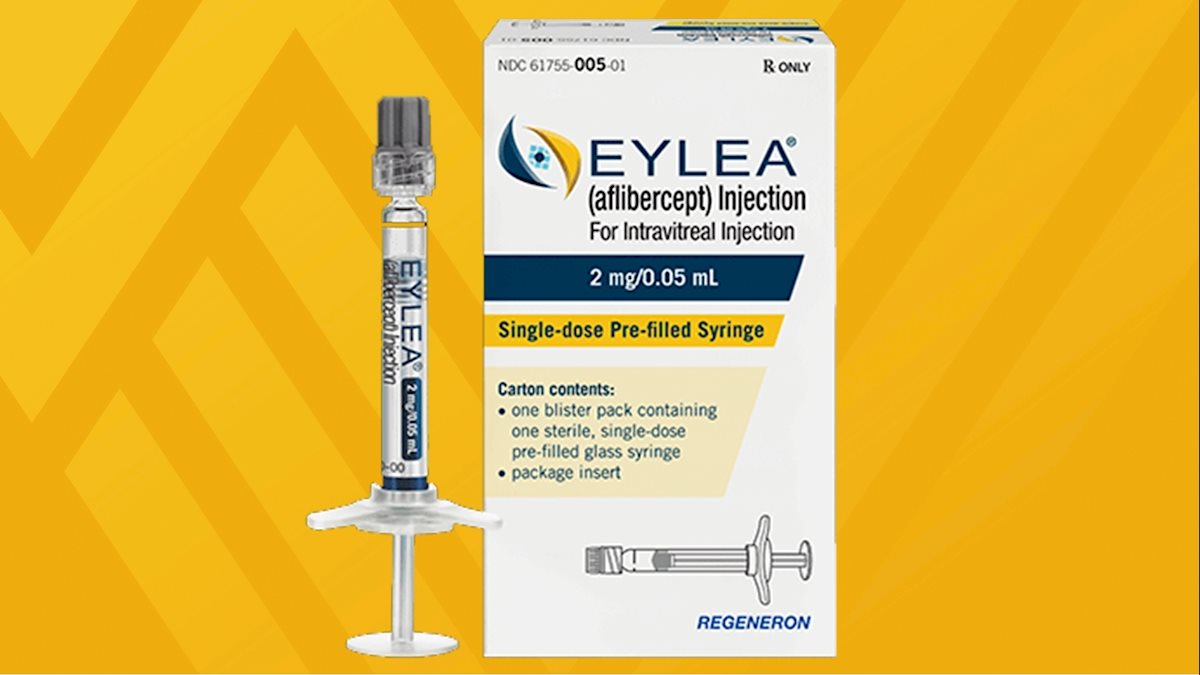
EYLEA 40 mg/mL (aflibercept solution for intravitreal injection): Higher risk of intraocular pressure increase with the pre-fill